When you're on multiple medications that each have a razor-thin line between working and causing harm, switching to a generic version isn't just a cost-saving move-it can be life-threatening. This is the reality for patients taking combination NTI drugs: two or more Narrow Therapeutic Index medications used together. These aren't your everyday pills. They're the kind where a 5% change in blood levels can mean the difference between control and crisis. And yet, while single NTI drugs like warfarin or levothyroxine have generic versions, almost none of their combination forms do. Why? And what does that mean for patients who need them?
What Exactly Are NTI Drugs?
NTI stands for Narrow Therapeutic Index. These are drugs with a tiny window between the dose that works and the dose that poisons you. Think of it like walking a tightrope blindfolded. Too little, and the treatment fails. Too much, and you end up in the hospital.
The FDA defines NTI drugs by five key traits:
- Therapeutic and toxic doses are barely separated
- Small changes in blood levels can cause serious harm
- They often require regular blood tests to monitor levels
- Patients usually need frequent dose adjustments
- There’s very little variation in how one person processes the drug compared to another
Common examples include warfarin (a blood thinner), levothyroxine (for thyroid issues), lithium (for bipolar disorder), digoxin (for heart rhythm), phenytoin (for seizures), and carbamazepine (for nerve pain and epilepsy). Each of these has been studied for decades. But when you combine two of them-say, warfarin and amiodarone, both NTI drugs-the risks multiply.
Why Combination NTI Drugs Are So Hard to Make Generic
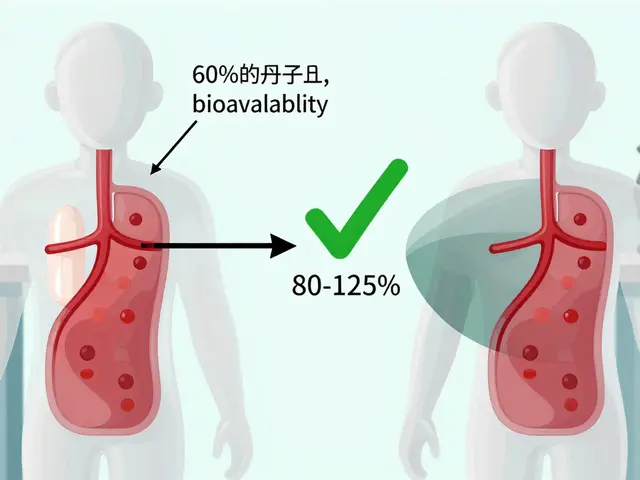
Generic drug approval isn't just about copying a pill. It’s about proving bioequivalence: that the generic version delivers the same amount of active ingredient into your bloodstream at the same rate as the brand-name drug. For regular drugs, the standard is 80% to 125% similarity in absorption (measured by AUC and Cmax). But for NTI drugs, the bar is much higher.
Since 2022, the FDA requires generics of single NTI drugs to match within 90.00% to 111.11% for Cmax and 90.00% to 112.00% for AUC. That’s a much tighter range. But when a combination contains two NTI drugs? The challenge explodes.
Imagine two drugs, each with a 10% allowable variation. When taken together, the total variation isn’t 10%-it’s closer to 20%. And if each component drifts in the same direction (say, both slightly higher), the risk of overdose skyrockets. If one drifts higher and the other lower, the treatment fails. There’s no way to predict this reliably in lab tests.
As a result, no fixed-dose combination containing two NTI drugs is approved in the U.S. market. While there are 11 generic versions of warfarin alone, there are zero generics of warfarin plus another NTI drug. Same with lithium plus valproate. Same with any other combo. The science just hasn’t caught up.
What Happens When Patients Switch to Generics?
Even when a combination includes just one NTI drug, problems arise. A 2020 study in JAMA Internal Medicine found that patients on combination therapies with one NTI drug had a 27% higher rate of adverse events after switching to generics, compared to just 8% for non-NTI combinations.
Real-world stories back this up. One patient on a warfarin-amiodarone combo posted on a health forum: “My INR went from 2.5 to 6.8 in three days after my pharmacy switched to generic warfarin. I ended up in the ER with internal bleeding.” That’s not rare. A 2023 survey of 856 pharmacists found that 78.3% had seen treatment failure after generic substitution in NTI combinations. Over 40% reported serious adverse events.
And it’s not just about safety. It’s about stability. Patients on combination NTI therapy need 3.2 dose adjustments on average, compared to 1.8 for non-NTI regimens. That means more blood tests, more doctor visits, more stress. Annual monitoring costs for these patients average $1,200-$2,500-three times more than for non-NTI combinations.

Who’s Saying What?
Experts are divided. The American Society of Health-System Pharmacists (ASHP) issued a position statement in 2021 opposing automatic substitution of generic NTI drugs in combination regimens, calling the risk “unacceptable.” Dr. Aaron Kesselheim from Harvard showed that 18.7% of patients on generic warfarin had unstable INR levels, versus just 4.3% on brand-name.
But it’s not all doom. Generic manufacturers like Teva and Sandoz point to Europe, where levothyroxine combinations have been used safely since 2015 with less than 2% adverse events. The European Medicines Agency (EMA) implemented similar tight bioequivalence standards as the FDA in early 2023.
Still, Dr. Donald Berry, a biostatistician at MD Anderson, put it bluntly in Nature Reviews Drug Discovery: “The 90-111% window for NTI drugs still allows 22% total variation. When you combine two such drugs, that’s a 44% total variation risk. That’s not just statistically significant-it’s clinically dangerous.”
The Coverage Gap: Who’s Left Behind?
Here’s the cold truth: the U.S. market for NTI drugs was worth $48.7 billion in 2023. But combination NTI products? They make up less than 0.3% of that market. Why? Because no one can get them approved.
While 87.4% of single NTI drugs have generic versions, only 12.6% of combination regimens involving NTI drugs do. And most of those are outside the U.S. In Australia, Canada, and parts of Europe, some combinations are available-but not here. That means American patients on these regimens are stuck with expensive brand-name drugs. Or worse, they’re switched to separate generics, forcing them to manage multiple pills, different dosing schedules, and more chances for error.
Enterprise healthcare systems are 3.2 times more likely than community pharmacies to block automatic substitution of NTI combinations. Why? Because they’ve seen the outcomes. Emergency visits. ICU admissions. Deaths.

What’s Next?
The FDA released draft guidance in February 2023 proposing even stricter standards for combination NTI drugs: 90.00%-107.69% for Cmax and 90.00%-110.00% for AUC. That’s tighter than any current standard in the world. It’s a step forward, but it’s not enough. The real solution may lie in something called “precision bioequivalence”-using computer modeling to simulate how drugs interact in real patients, not just in a lab.
The FDA plans a pilot program in 2024 to test this approach. If it works, it could open the door to combination NTI generics. But even optimists admit it’s years away. Dr. Robert Temple, former FDA official, believes manufacturing advances may eventually make it possible. Dr. Lewis Nelson at NYU isn’t so sure. “Combining two narrow-window drugs,” he said, “is like stacking two unstable towers. No matter how good your bricks are, the whole thing still falls.”
What Should Patients Do?
If you’re on a combination therapy that includes one or more NTI drugs:
- Ask your pharmacist: Is this a brand or generic? Is it a fixed-dose combo or separate pills?
- Don’t let your pharmacy switch medications without your doctor’s approval.
- Keep detailed records of your blood test results and any symptoms.
- Advocate for staying on brand-name if you’re stable-your safety isn’t worth the savings.
- Check if your provider offers a specialized NTI clinic. Only 12 of 50 major U.S. hospitals do.
There’s no easy fix here. The science, the regulation, the economics-they’re all misaligned. For now, the safest path is clear: avoid switching unless you’re under close, expert supervision. Because when the margin for error is this small, you can’t afford to gamble.



Buddy Nataatmadja
March 13, 2026 AT 21:02mir yasir
March 15, 2026 AT 02:41Jinesh Jain
March 16, 2026 AT 03:19douglas martinez
March 17, 2026 AT 22:49Katherine Rodriguez
March 19, 2026 AT 13:07Devin Ersoy
March 19, 2026 AT 14:19Scott Smith
March 20, 2026 AT 03:20Sally Lloyd
March 22, 2026 AT 00:35Emma Deasy
March 23, 2026 AT 13:42tamilan Nadar
March 24, 2026 AT 02:28Adam M
March 25, 2026 AT 18:48Rosemary Chude-Sokei
March 26, 2026 AT 08:07