You pick up your prescription and notice the pill is blue and round, but last month it was white and oval. It’s a jarring feeling. You might wonder if you've been given the wrong medication or if this new version actually works. The truth is, when you switch between generic manufacturers is pharmaceutical companies that produce non-branded versions of medications after patents expire, the look of the pill almost always changes, even if the medicine inside is identical. Understanding why this happens and how to verify your medication can save you from unnecessary stress and pharmacy trips.
Why Your Generic Pills Look Different
It is a common misconception that generic drugs should look exactly like the brand-name version. In reality, trademark laws actually prevent generic manufacturers from copying the exact color, shape, and markings of the original brand. If a company made a generic that looked identical, they could be sued for trademark infringement.
Because of this, manufacturers use different inactive ingredients-fillers, binders, and dyes-to create their own unique version. According to a 2020 study in Nature Scientific Reports, color differences occur in about 78% of generic variations, while shape differs in 65% and size in 42%. This means that if your pharmacy switches from one manufacturer (like Teva) to another (like Sandoz), your pill's appearance will likely change, but the active drug remains the same.
Understanding Bioequivalence: Does it Work the Same?
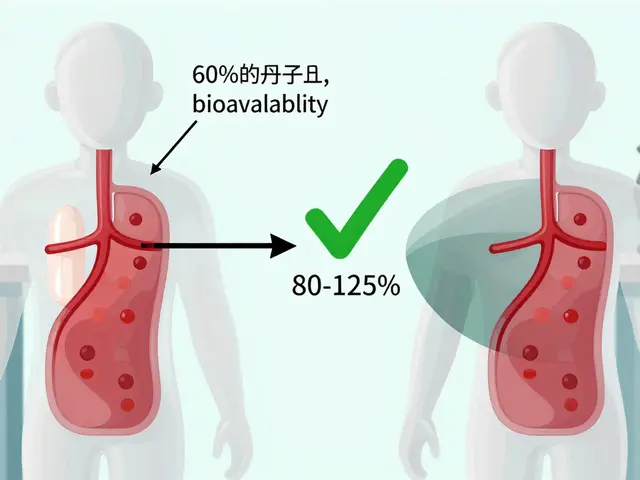
The core requirement for any generic is bioequivalence, which is the demonstration that a generic drug delivers the same amount of active ingredient into the bloodstream at the same rate as the brand-name drug. The FDA (U.S. Food and Drug Administration) mandates that generics be therapeutically equivalent. This means they must have the same active ingredient, strength, dosage form, and route of administration.
For most drugs, the FDA allows a confidence interval of 80-125% for the geometric mean ratios of the drug's concentration in the blood. While that sounds like a wide gap, a landmark study in the Journal of Generic Medicines found the average actual difference in absorption was only 3.5%-which is often less than the variation found between two different batches of the brand-name drug itself.
| Feature | Standard Generic | NTID (e.g., Warfarin, Levothyroxine) |
|---|---|---|
| Bioequivalence Range | 80-125% | Tighter range (90-111%) |
| Switching Risk | Very Low | Moderate (May require monitoring) |
| Manufacturer Stability | Flexible substitution | Recommended to stay with one maker |

How to Compare and Verify Your Medication
If you are unsure about a pill's appearance, you don't have to guess. There are professional tools used by pharmacists and patients to ensure the right drug is being taken. Every prescription pill is required to have a unique imprint-a combination of letters and numbers-that acts as a fingerprint for that specific manufacturer's version of the drug.
Here is a practical way to verify your medication:
- Check the Imprint: Look at the letters or numbers stamped onto the pill. This is the most reliable way to identify the drug.
- Use a Pill Identifier: Tools like the Drugs.com Pill Identifier allow you to input the color, shape, and imprint to see exactly which manufacturer produced that pill.
- Consult the Orange Book: The FDA Orange Book is the official gold standard for checking therapeutic equivalence. It lists which generics are approved substitutes for which brand drugs.
- Verify the NDC: Every drug has a National Drug Code (NDC), a unique 10 or 11-digit number that identifies the manufacturer, the product, and the package size. You can find this on your prescription bottle.
When Appearance Changes Actually Matter
For the vast majority of people, a change in pill color or shape is harmless. However, there is a specific category called Narrow Therapeutic Index (NTI) drugs. These are medications where a tiny difference in dosage or absorption can lead to a significant change in how the drug works or cause toxicity.
Common examples include warfarin (a blood thinner) or levothyroxine (for thyroid issues). For these specific drugs, switching between generic manufacturers can sometimes cause fluctuations in blood levels. This is why the American Medical Association suggests staying with the same generic maker for NTI drugs unless a change is medically necessary. If you are on one of these, you can ask your doctor for a "Dispense as Written" (DAW) instruction, which tells the pharmacist not to substitute the manufacturer.

The Role of Major Generic Manufacturers
Not all generic companies are the same. Some focus on simple tablets, while others specialize in "complex generics" like inhalers or injectables, which take much longer to develop and approve. Global giants like Teva Pharmaceutical Industries and Sandoz dominate the market through massive scale and operational efficiency.
When you see a price drop in your medication, it's usually because more of these competitors have entered the market. Data shows that prices can plummet by 70-80% once ten or more manufacturers start producing the same generic. While this is great for your wallet, it's the reason why your pharmacy might switch your pill's appearance frequently-they are often switching to the most cost-effective manufacturer available.
Is it safe to switch generic manufacturers every month?
For most medications, yes. Because of strict bioequivalence standards, different generics of the same drug will perform nearly identically in your body. However, if you are taking a Narrow Therapeutic Index (NTI) drug, you should consult your doctor before switching, as small variations can impact your health.
Why doesn't my generic look like the brand-name pill?
Trademark laws prevent generic companies from copying the exact look of a brand-name drug. They must create their own unique shape, color, and imprint to avoid legal issues, even though the active medicine is the same.
How can I tell if I have the right medication if the pill looks different?
Check the imprint code on the pill and match it using a reliable pill identifier tool or by asking your pharmacist. You can also check the NDC number on your prescription label to verify the manufacturer.
Do inactive ingredients in generics cause side effects?
While the active ingredient is the same, the inactive ingredients (fillers and dyes) vary. In rare cases, a person may be allergic to a specific dye or filler used by one manufacturer but not another. If you notice a new allergy after a switch, notify your doctor.
What is the FDA Orange Book?
The Orange Book is the official FDA publication that lists all approved drug products with therapeutic equivalence evaluations. It allows healthcare providers to see which generic versions are considered interchangeable with the brand-name drug.
Next Steps for Patients
If you are concerned about your medication's appearance, start by documenting it. Take a photo of your pill and the imprint code. When you go for your next refill, if the pill looks different, ask your pharmacist: "Which manufacturer produced this version, and is it bioequivalent to the last one I had?"
For those on critical medications like blood thinners or thyroid hormones, ask your provider to write "Dispense as Written" or a specific manufacturer's name on the prescription. This prevents the pharmacy from rotating manufacturers and ensures your dosing remains consistent across every refill.



Sam Hayes
April 4, 2026 AT 16:22just a heads up for everyone that the ndc number is your best friend here. if you feel weird about a switch just ask the pharmacist to show you the bottle they are filling from it clears up any confusion immediately
Joseph Rutakangwa
April 6, 2026 AT 10:05pill identifiers are great tools for peace of mind
Will Baker
April 8, 2026 AT 04:12oh wow look at us trusting the government and big pharma just because a little chart says it is okay. i bet those bioequivalence ranges are just made up to keep us from complaining while the pharmacies play musical chairs with our health for a few cents profit. it is almost cute how people actually believe a pill identifier proves anything when the whole system is designed to confuse you
Rob Newton
April 8, 2026 AT 09:55bioequivalence is a myth. the nuances matter
The Charlotte Moms Blog
April 10, 2026 AT 01:51Actually!!! The inactive ingredients can be a total nightmare for some people... Especially for kids... Some dyes cause behavioral issues!!! The post barely mentions the risks of fillers... It is honestly irresponsible to say switching is harmless for the vast majority without a warning about sensitivities!!!
Vicki Marinker
April 11, 2026 AT 02:05The formatting of this information is acceptable, but the premise is dreadfully optimistic. One simply cannot assume that the FDA's oversight is infallible. It is quite tedious to see such blind faith in regulatory bodies placed in a public forum. The reality of pharmaceutical manufacturing in global hubs is often far less stringent than the Orange Book would suggest. I find the suggestion to use a website to verify medication rather than insisting on a specific brand to be a lazy approach to healthcare. It is a pity that patients are encouraged to be their own pharmacists.
Dee McDonald
April 12, 2026 AT 13:08Get on top of your health people! If you are on one of those NTI drugs, do not just sit there and let the pharmacy swap your meds! Push your doctor for that DAW order right now! It is your body and your life on the line! Why should we accept a 10% variance in something as critical as thyroid hormone? It is time to be aggressive about our prescriptions and demand consistency regardless of the cost to the insurance company! Go check your pill imprints today and start a log!
Sakshi Mahant
April 14, 2026 AT 03:27It is interesting to see how different countries handle generic labeling. In some places, the transparency is a bit different, but the goal of affordable medicine is a universal bridge that helps so many people get the care they need.
HARSH GUSANI
April 16, 2026 AT 00:00This is all just US talk! 🇮🇳 In India we have amazing generic medicines that are way better and cheaper than these expensive Western brands! You people are too scared of a blue pill vs a white pill while we lead the world in pharmaceutical exports! 💊💪 The FDA is just trying to protect their own companies by making everyone afraid of generics! Stop listening to the fear and trust the science of the East! 🇮🇳✨
Lawrence Rimmer
April 17, 2026 AT 08:58The duality of man is basically encapsulated in the fear of a changing pill color. We crave the chemical effect but dread the physical manifestation of the delivery system. It is all a simulation of stability in an unstable world anyway. The pharmacy is just a temple of modern alchemy where we trade gold for colored stones. Honestly it is just a chore to even read about it
Hudson Nascimento Santos
April 18, 2026 AT 13:34There is a certain philosophical tension here between the empirical data of bioequivalence and the subjective experience of the patient. When the physical form of a medicine changes, it challenges our psychological trust in the cure. We are not just consuming molecules; we are consuming the concept of reliability. If the pill changes, the ritual of healing is interrupted, and that is where the anxiety stems from, regardless of whether the active ingredient is identical.