When you pick up a prescription at the pharmacy, you might be handed a generic pill instead of the brand-name version you asked for. It’s cheaper, sure - but is it the same? This question comes up often, especially when you’re managing a chronic condition. The answer lies in two closely related but very different concepts: bioavailability and bioequivalence. Understanding the difference between them helps you know why generics are safe, when they might not be, and how regulators make sure they work just like the original.
What Bioavailability Really Means
Bioavailability is about how much of a drug actually gets into your bloodstream after you take it. Think of it as the percentage of the pill that makes it past your stomach and liver to do its job. Not all of it does. Some gets broken down during digestion. Some gets filtered out by your liver before it even reaches your blood. This is called first-pass metabolism.
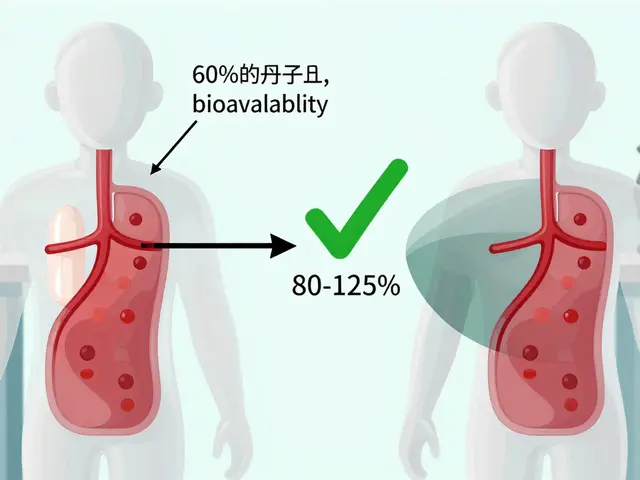
For example, if you take a 100 mg tablet and only 60 mg ends up in your blood, that drug has 60% bioavailability. If you get the same drug as an injection, nearly 100% enters your bloodstream right away. That’s why intravenous (IV) drugs are used as the gold standard - they’re 100% bioavailable.
For oral drugs, bioavailability depends on three things: how well the pill dissolves in your gut, how well your gut absorbs it, and how much your liver breaks down before it circulates. A drug with low bioavailability might need a higher dose to work. That’s why some medications come in different strengths or forms - like extended-release pills or liquid versions.
What Bioequivalence Is and Why It Matters
Bioequivalence is not about one drug. It’s about comparing two. Specifically, it’s how regulators prove that a generic drug performs the same as the brand-name version. It’s not enough for a generic to have the same active ingredient. It must deliver that ingredient to your body at the same rate and in the same amount.
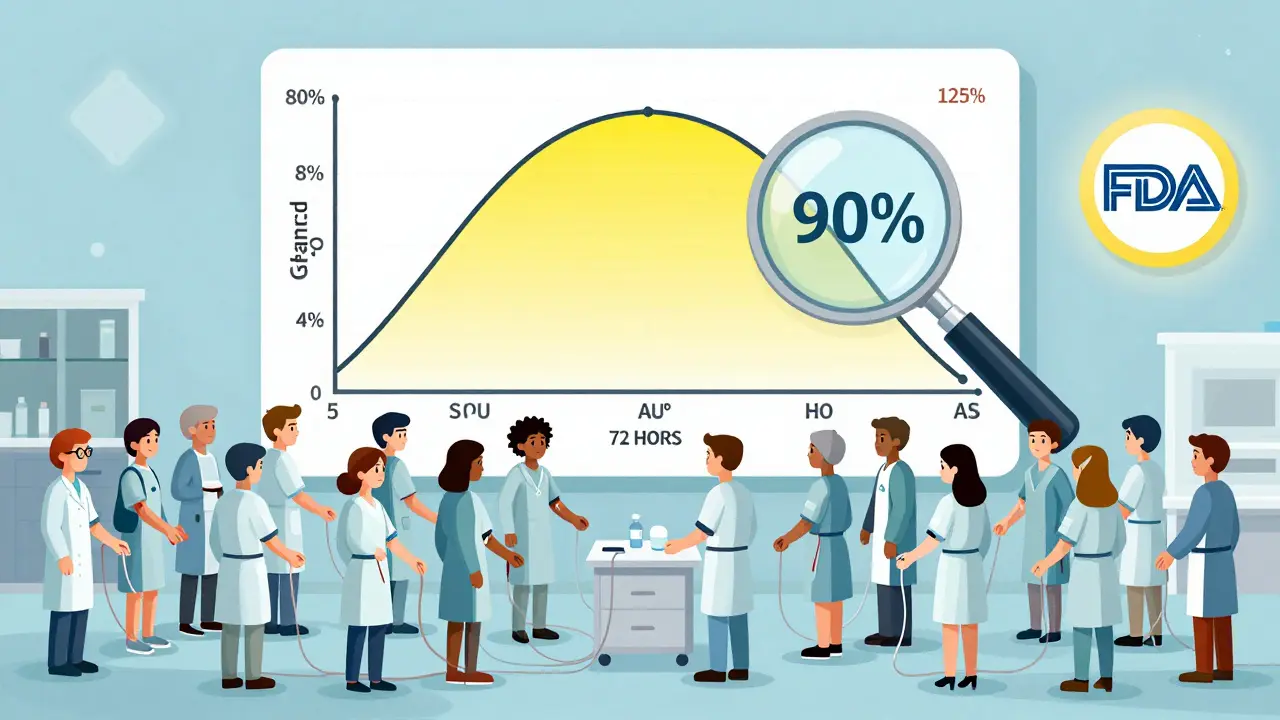
The FDA and other global agencies require a head-to-head study. Healthy volunteers take both the brand-name drug (the reference) and the generic (the test) under controlled conditions - usually fasting, in a clinical setting. Blood samples are taken over 72 hours to track how the drug moves through the body.
The two most important numbers they look at are:
- AUC - the total amount of drug in your blood over time (area under the curve). This tells you how much of the drug you’re exposed to overall.
- Cmax - the highest concentration reached in your blood. This tells you how fast the drug gets absorbed.
For two products to be declared bioequivalent, the 90% confidence interval of the ratio between the generic and brand must fall between 80% and 125%. This is called the 80/125 rule. It means the generic can’t be more than 20% weaker or stronger than the original in terms of total exposure or peak concentration.
This rule isn’t arbitrary. It’s based on decades of data showing that differences smaller than this rarely affect how well a drug works or whether it causes side effects. But there’s a catch: for drugs with a narrow therapeutic index - where the difference between a safe dose and a toxic one is very small - the rules are tighter. For example, warfarin (a blood thinner) requires a range of 90-112% for AUC. Too wide, and you risk clotting or bleeding.
The Key Difference: One vs Two
Bioavailability describes a single drug. It answers: How well does this pill work? Bioequivalence compares two. It answers: Is this generic just as good as the brand?
You can measure bioavailability for any formulation - a new capsule, a chewable tablet, a liquid. But bioequivalence only makes sense when you’re comparing. A generic manufacturer doesn’t just say, “Our drug has 75% bioavailability.” They say, “Our drug has 98% relative bioavailability compared to the brand.” That’s the proof regulators need.
That’s also why you can’t assume two generics are bioequivalent just because they’re both generics. Each one must be tested against the original brand-name drug. A generic made by Company A might be bioequivalent to the brand, but a different generic from Company B might not be bioequivalent to Company A’s version - even though both are generics. They’re not tested against each other.
Real-World Evidence: Do Generics Work?
Some people worry that generics don’t work as well. But the data says otherwise. Between 2010 and 2020, 99.7% of generic drugs approved by the FDA met bioequivalence standards. That’s nearly perfect.
Dr. Sarah Chen, a pharmacist in Ohio, tracked 1,247 patients switched from brand to generic blood pressure medications. Only 17 (1.36%) reported issues. Of those, only 4 cases (0.32%) were confirmed as true therapeutic failures - and even those were linked to other factors like non-adherence or changing health conditions.
Still, there are exceptions. Levothyroxine, used for thyroid conditions, is one of the most common drugs where patients report differences. Why? Because thyroid hormone levels are extremely sensitive. Even small variations in absorption can throw off lab results. The FDA requires stricter bioequivalence standards for levothyroxine than for most other drugs - and still, some patients report problems. That’s why many doctors stick to one brand or generic and avoid switching.
A 2022 survey by Patients for Better Drugs found that 87.4% of people saw no difference after switching to generics. The remaining 12.6% reported issues - but only 3.8% of those cases were medically confirmed as bioequivalence-related. Most were placebo effects, anxiety, or unrelated health changes.
How These Studies Are Done
Bioequivalence studies aren’t simple. They follow strict rules. Here’s what happens:
- 24 to 36 healthy volunteers are enrolled.
- They take the brand drug in one period, then the generic in another - usually with a washout period in between.
- They fast overnight before each dose. Food can change absorption, so studies are done under controlled conditions.
- Blood is drawn 12 to 18 times over 72 hours.
- The samples are analyzed using highly sensitive lab equipment to measure exact drug concentrations.
- Data is analyzed statistically to see if the 80-125% range is met.
It takes 3 to 6 months to design and run one of these studies. The cost? Often over $500,000. That’s why only large manufacturers can afford to make generics - and why complex drugs (like inhalers or injectables) are harder and more expensive to copy.
When Bioequivalence Isn’t Enough
Not all drugs are created equal. Some are tricky to test.
Topical creams, nasal sprays, and eye drops don’t enter the bloodstream the same way pills do. Their effectiveness depends on local tissue absorption - not blood levels. That’s why regulators are developing new methods, like in vitro testing (lab tests that simulate skin or mucous membrane absorption).
Another challenge is food effects. Some drugs, like voriconazole (an antifungal), absorb 36% more Cmax when taken with a high-fat meal. That means a generic tested on an empty stomach might not behave the same way if the patient eats with it. So, regulators now require both fasting and fed-state studies for certain drugs.
And then there’s the future. By 2027, experts predict that 30% of bioequivalence assessments for complex drugs will use computer modeling - called PBPK (physiologically-based pharmacokinetic) modeling. These models simulate how a drug moves through the body based on anatomy, metabolism, and physiology. It’s not a replacement for human studies, but it helps predict outcomes faster and cheaper.
Final Takeaway: Trust the Science, Stay Informed
Bioavailability and bioequivalence aren’t just regulatory buzzwords. They’re the foundation of safe, affordable medicine. Generics aren’t “second-rate.” They’re scientifically proven alternatives that save patients and healthcare systems billions.
But no system is perfect. For most drugs - antibiotics, statins, antidepressants - generics work just as well. For others - thyroid meds, blood thinners, seizure drugs - switching brands or generics can sometimes cause issues. That’s why your doctor or pharmacist might recommend staying on the same version.
If you’re switching from brand to generic and notice changes - feeling different, side effects, or lab results shifting - talk to your provider. It’s not always the drug. But it’s worth checking.
Are generic drugs as effective as brand-name drugs?
Yes, for the vast majority of drugs, generics are just as effective. The FDA requires that generics meet strict bioequivalence standards, meaning they deliver the same amount of active ingredient at the same rate as the brand. Between 2010 and 2020, 99.7% of approved generics met these criteria with no significant therapeutic differences.
Why do some people say generics don’t work for them?
In most cases, the issue isn’t bioequivalence - it’s other factors. Patients may notice changes in pill size, color, or inactive ingredients (like fillers), which can trigger placebo effects. Some drugs, like levothyroxine or warfarin, have a narrow therapeutic window, meaning tiny changes in absorption can matter. But studies show only about 0.3-0.5% of cases are true therapeutic failures linked to bioequivalence.
Is bioequivalence the same in every country?
Most major regulators - the FDA, EMA (Europe), Health Canada, and others - use the same 80-125% range for bioequivalence. This is thanks to global harmonization through the ICH. However, some countries may have stricter rules for certain drugs, especially those with narrow therapeutic indexes. The core science is the same worldwide.
Can two different generics be bioequivalent to each other?
No. Bioequivalence is always tested against the original brand-name drug (the reference listed drug). Two generics may both be bioequivalent to the brand, but they are not tested against each other. So, switching between two different generics could theoretically lead to differences - which is why doctors often recommend sticking with one manufacturer.
What happens if a generic fails bioequivalence testing?
It’s not approved. If the 90% confidence interval for AUC or Cmax falls outside the 80-125% range, the FDA rejects the application. The manufacturer must revise the formulation, run new studies, and resubmit. This is rare - most failures happen early in development. Once approved, the generic can’t be changed without retesting.



Melissa Stansbury
March 18, 2026 AT 17:06I switched from brand-name lisinopril to a generic last year and my blood pressure went haywire for two weeks. My doctor said it was "probably placebo," but I swear the pills felt different. Same active ingredient, same dosage-same box even-but something was off. I went back to the brand and boom, stable again. Not saying generics are bad, but I don’t trust them anymore. If your life depends on it, why gamble?
cara s
March 20, 2026 AT 10:28It is absolutely fascinating, and I must say, profoundly concerning, that the regulatory framework surrounding bioequivalence is built upon such a narrow statistical window-80% to 125%-when we are dealing with pharmacokinetic profiles in human subjects who vary wildly in metabolism, gut flora, and hepatic enzyme activity. The assumption that this range is universally safe ignores the fact that some individuals are ultra-rapid metabolizers, while others are slow, and for drugs with a narrow therapeutic index, even a 10% deviation can precipitate clinical catastrophe. I am not a medical professional, but I have read the literature, and I find it alarming that this standard persists without individualized pharmacogenomic testing being mandated.
Amadi Kenneth
March 20, 2026 AT 21:45They say generics are safe... but who's really behind this? Big Pharma invented generics to keep you hooked-sell you the brand for $200, then when patents expire, they sell the generic for $5 under a different name... same factory, same workers, same machine. You think the FDA checks every batch? Nope. They sample 1 in 500. And the labs? Mostly outsourced to China and India. I've seen the reports. Contaminants. Inconsistent dissolution. You think your thyroid med is working? Maybe it's just not killing you yet.
Shameer Ahammad
March 22, 2026 AT 16:52Let me be perfectly clear: the notion that bioequivalence is a reliable proxy for therapeutic equivalence is not only scientifically flawed-it is ethically indefensible. The 80-125% rule is a political compromise, not a biological truth. For a drug like warfarin, where a 5% variation in AUC can lead to stroke or hemorrhage, this is not merely negligent-it is criminal. And yet, we allow pharmacies to switch patients without consent, without notification, without informed consent. This is not healthcare. This is cost-cutting masquerading as progress. We need mandatory therapeutic drug monitoring for all patients on generics. Period.
Alexander Pitt
March 22, 2026 AT 20:11The data is overwhelming: 99.7% of generics meet bioequivalence standards. That’s not luck. That’s rigorous science. The few cases where people report issues are almost always due to placebo, changes in inactive ingredients, or underlying health shifts-not bioequivalence failure. If you’re on levothyroxine or warfarin, stick with one manufacturer. But don’t let anecdotal fear override evidence. Generics save millions of lives and billions in healthcare costs. The system works. Trust the process.
Manish Singh
March 23, 2026 AT 13:28In India, we've been using generics for decades. My grandmother takes her antihypertensives as generics-and she’s 82, still gardening, still cooking, still sharp. The fear of generics is mostly a Western luxury. Here, people choose generics because they have to. And guess what? They live. The science is solid. The regulators may be imperfect, but the outcome? Millions of people getting life-saving meds they couldn’t afford otherwise. Don’t let fear silence access.
Nilesh Khedekar
March 24, 2026 AT 02:13Okay but like... have you ever seen the pills? I switched from brand to generic and the generic had this weird chalky taste? Like, I swear it was different. And my anxiety got worse. Coincidence? I don't think so. Also, why do they use lactose in some and cornstarch in others? I'm gluten intolerant and I didn't even know till I got sick. They don't even tell you! And the FDA? They don't care. It's all about money. Big Pharma owns the system. I'm not paranoid-I'm informed.
Robin Hall
March 24, 2026 AT 15:25The 80-125% bioequivalence standard is a dangerous fiction. It assumes homogeneity in human physiology that simply does not exist. Genetic polymorphisms in CYP450 enzymes alone create variability far beyond this range. Furthermore, the use of healthy volunteers-typically young, lean, non-smokers-is a gross misrepresentation of the actual patient population: elderly, comorbid, polypharmacy users. This is not science. It is a regulatory charade designed to protect corporate profits under the guise of public health. The FDA's approval process for generics is a sham.
Suchi G.
March 24, 2026 AT 19:01I just want to say… I cried when I had to switch from my brand-name antidepressant to a generic. I felt like I lost a part of myself. My mood tanked. I couldn’t sleep. I thought I was failing again. I went back to my doctor and begged for the brand. They said it was "all in my head." But I know better. My body knew. The pills looked different. Smelled different. Even felt different in my throat. I don’t care what the stats say. If it breaks you, it’s not working. I’m not a number. I’m a person. And I deserve to feel stable.
becca roberts
March 26, 2026 AT 06:53So let me get this straight: we’ve got a system where two generics can both be "bioequivalent" to the brand, but not to each other… and we’re okay with patients being switched between them like a game of musical pills? That’s not science. That’s a corporate loophole dressed up as public policy. Also, who decided that 80-125% is "safe"? Did they test this on a 70-year-old with kidney disease? Or just on 24-year-old college kids who’ve never taken more than ibuprofen? I’m not impressed.
Andrew Muchmore
March 27, 2026 AT 13:05Generics work. The data says so. If you have issues, talk to your pharmacist. Don’t blame the system.
Paul Ratliff
March 28, 2026 AT 14:28my cousin in delhi takes 5 diff generics for his bp and he’s fine. usa overthinks everything. just take the pill.
SNEHA GUPTA
March 30, 2026 AT 04:11There’s a deeper philosophical question here, isn’t there? When we reduce a human being’s health to a statistical range-80% to 125%-we are not merely measuring drug absorption. We are quantifying the value of human life. Are we comfortable with the idea that someone’s well-being is deemed "sufficient" if it falls within a margin of error defined by regulatory bureaucrats? Bioequivalence is a technical achievement. But is it ethical? Are we prioritizing cost over care? Or worse-are we normalizing the idea that some lives are worth less because their medication must be cheaper?
Gaurav Kumar
March 30, 2026 AT 07:08USA thinks it invented medicine. We in India have been making high-quality generics for 50 years. The world uses our drugs. The WHO trusts us. The FDA? They inspect our factories. They approve them. And yet, Americans still fear generics like they’re some kind of conspiracy. It’s not the drug-it’s your arrogance. We make better generics than your brand-name stuff. And we do it for 1/10th the price. Stop being scared. Start being proud.