Tag: bioequivalence

How to Compare Generic Drug Manufacturers and Pill Appearance
Learn how to compare generic drug manufacturers and why pill appearance changes. Understand bioequivalence, NTI drugs, and how to verify your medication using the FDA Orange Book.
April 4 2026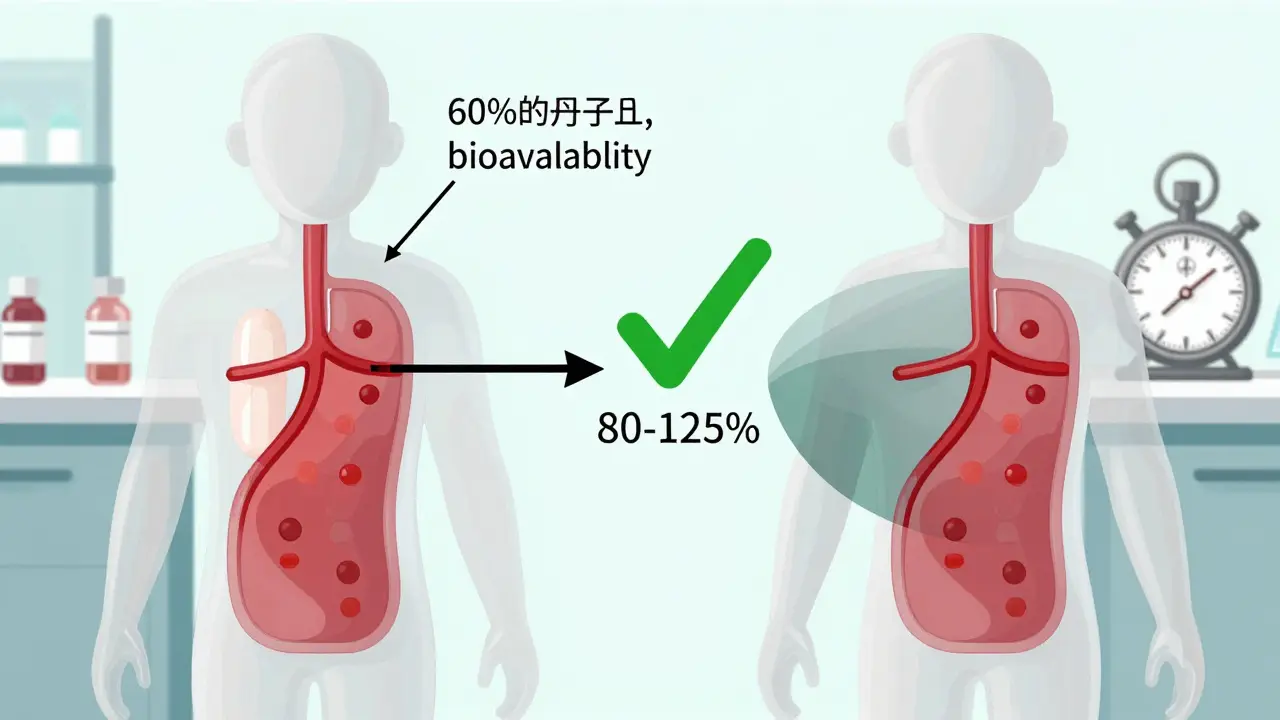
How to Compare Bioavailability and Bioequivalence in Generic vs Brand Drugs
Bioavailability measures how much of a drug enters your bloodstream, while bioequivalence proves a generic matches the brand. Learn how regulators ensure generics work just as well - and when exceptions apply.
March 17 2026
Combination NTI Drugs and Generic Availability: Coverage and Gaps
Combination NTI drugs offer critical treatment for complex conditions, but generic versions are nearly nonexistent due to strict bioequivalence challenges. Patients face higher risks of failure and toxicity when substitutions occur.
March 12 2026
Why Patients Prefer Brand-Name Medications Over Generics: Explained
This article explains why patients often prefer brand-name medications over cheaper generics despite identical effectiveness. It covers factors like perceived quality, trust, marketing influence, demographic trends, and real-world data from studies and surveys.
February 5 2026