© 2026. All rights reserved.
Tag: brand drugs
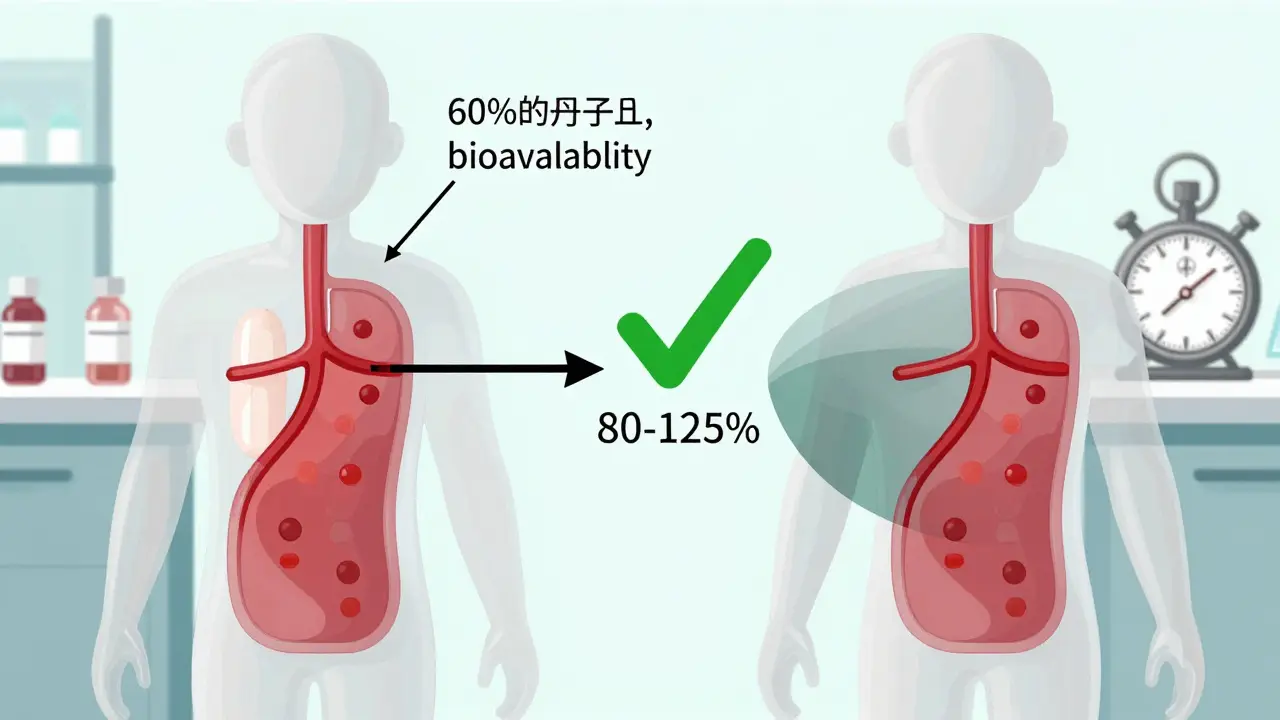
bioavailability
bioequivalence
generic drugs
brand drugs
pharmacokinetics
How to Compare Bioavailability and Bioequivalence in Generic vs Brand Drugs
Bioavailability measures how much of a drug enters your bloodstream, while bioequivalence proves a generic matches the brand. Learn how regulators ensure generics work just as well - and when exceptions apply.
March 17 2026