When a brand-name drug loses its patent, the race to bring down prices begins. But not all generics enter the market the same way. Two very different players show up: the first generic and the authorized generic. Their timing isn’t just a coincidence-it’s a calculated move that shapes how much you pay for your medicine.
What Is a First Generic?
A first generic is the very first company to successfully challenge a brand-name drug’s patent and get FDA approval to sell a generic version. This isn’t easy. It takes years of legal work, clinical testing, and regulatory filings. Under the Hatch-Waxman Act of 1984, the first company to file an Abbreviated New Drug Application (ANDA) gets 180 days of exclusive rights to sell the generic. During that time, no other generic can enter. This exclusivity was meant to reward the risk-taker-the company that spent millions fighting patents and proving bioequivalence.
That exclusivity used to mean big profits. A first generic could capture 70-90% of the market. For a drug like Lyrica (pregabalin), that meant hundreds of millions in revenue. Teva, for example, launched the first generic of Lyrica in July 2019 and expected to dominate the market. But what happened next changed everything.
What Is an Authorized Generic?
An authorized generic isn’t a challenger. It’s the brand-name company itself-or a partner it authorizes-selling the exact same drug under a generic label. No ANDA needed. No bioequivalence studies. Just a label change. These drugs are made in the same factory, with the same ingredients, and the same quality control as the brand. The only difference? The box says "pregabalin" instead of "Lyrica."
Here’s the twist: brand companies can launch an authorized generic at any time. They don’t have to wait for the patent to expire. They don’t even have to wait for the first generic to start selling. And they often time it perfectly.
Why Timing Matters
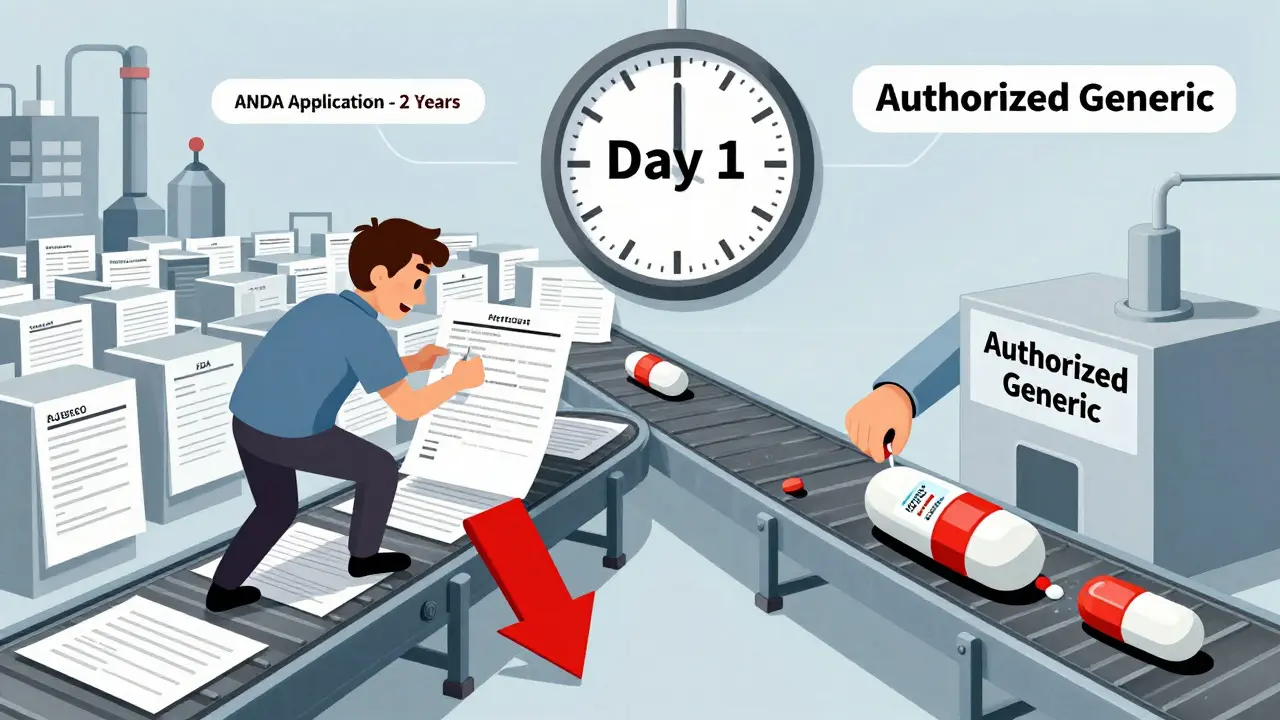
The real battle isn’t about who makes the drug-it’s about when they sell it. According to research from Health Affairs (2022), 73% of authorized generics hit the market within 90 days of the first generic’s approval. In 41% of cases, they launched on the exact same day.
This isn’t random. It’s strategy. When Teva launched its generic version of Lyrica, Pfizer didn’t wait. Within days, Pfizer’s own authorized generic, sold under Greenstone LLC, appeared on pharmacy shelves. Suddenly, instead of Teva owning 80% of the market, both companies split it. Teva’s revenue dropped by nearly half. Pfizer didn’t lose sales-they just changed the label and kept the profit.
That’s not competition. It’s a bypass. The 180-day exclusivity period was supposed to give the first generic a fair shot. But with an authorized generic already on the shelf, the market gets split before the first generic can build momentum.
The Financial Impact
Generic drugs usually slash prices by 80-90%. That’s the whole point. But when an authorized generic enters during the first generic’s exclusivity window, prices only drop 65-75%. That difference might sound small, but it adds up. The RAND Corporation estimates this tactic costs the U.S. healthcare system billions each year in lost savings.
For the first generic, the numbers are brutal. A typical first generic earns $100-500 million during its exclusivity period. But if an authorized generic enters early, that number can shrink to $45-300 million. For smaller generic manufacturers, that’s the difference between staying in business and shutting down.
The most affected drugs? Cardiovascular meds (32% of cases), CNS drugs like gabapentin and pregabalin (24%), and diabetes treatments (18%). Drugs like Eliquis and Jardiance are now battlegrounds where authorized generics are deployed as soon as the first generic appears.
How the System Was Designed vs. How It Works
The Hatch-Waxman Act was built on a simple idea: reward the first company to challenge a patent, and let competition drive prices down. But the system has been gamed.
Brand companies don’t need to wait for the patent to expire. They don’t need to go through the 10-month FDA review process. They can launch an authorized generic in days. Meanwhile, the first generic spends 2-3 years and $5-10 million on legal battles and regulatory filings, only to be undercut on day one.
The FDA approves about 80 first generics a year now, but less than 10% of generic applications clear the first review cycle. The backlog is real. Authorized generics? They skip the line entirely.
Who Wins? Who Loses?
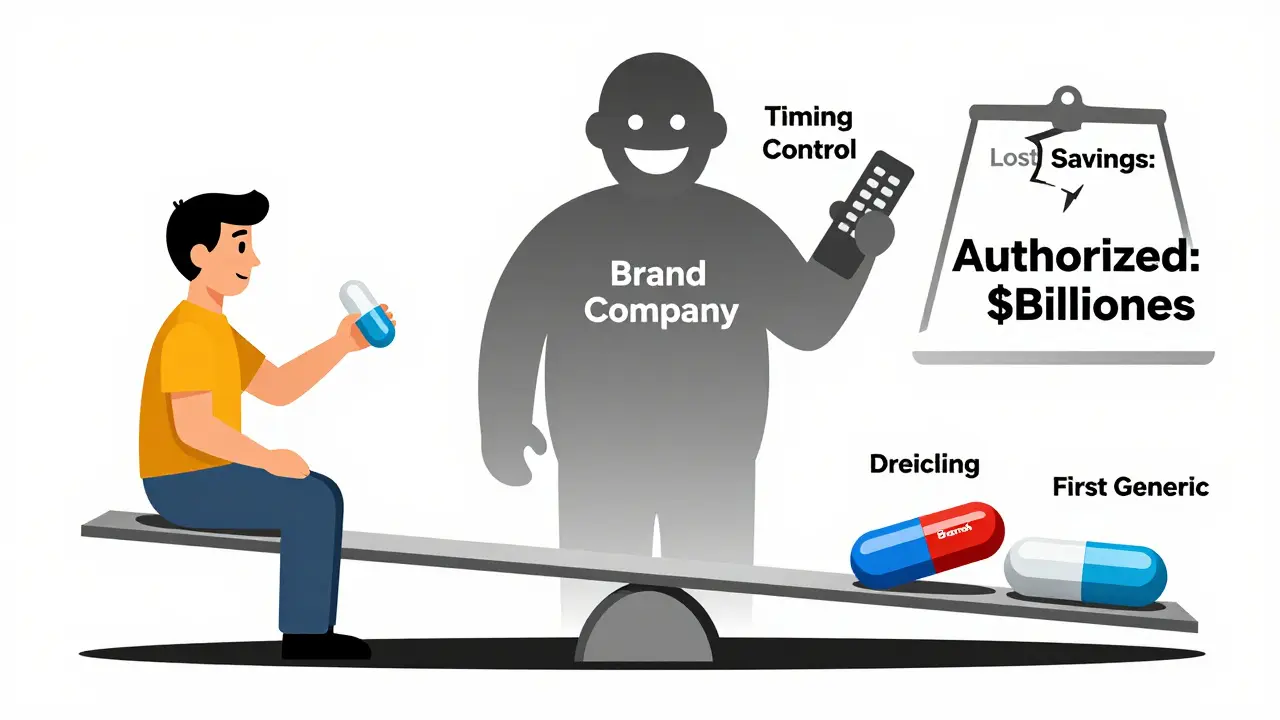
Brand companies win. They keep control of the market, avoid price collapse, and still collect revenue from the same product under a cheaper label.
Patients? They get a generic-but not the full price drop they should. A 65% reduction instead of 85% means higher out-of-pocket costs and more strain on insurers.
Smaller generic manufacturers? They’re the biggest losers. Many can’t afford to challenge patents if they know an authorized generic will appear the moment they win. Some have stopped trying altogether.
The Association for Accessible Medicines (AAM) argues that authorized generics still increase access and lower prices. They point to drugs like Lipitor and Prilosec, where generics brought down costs dramatically. But that’s not the full story. Those drugs didn’t have authorized generics launched on day one. The system worked because competition was real.
Today, that competition is often fake. The authorized generic isn’t a new entrant-it’s the brand in disguise.
What’s Changing Now?
The Inflation Reduction Act of 2022 took a clear stance: authorized generics are not considered "generic competitors" for Medicare price negotiation purposes. That’s a big deal. It means when the government tries to lower drug prices, it doesn’t count an authorized generic as real competition. That recognition is a step toward fixing the imbalance.
But enforcement is still weak. The FTC has challenged "pay-for-delay" deals where brand companies pay generics to delay entry. But when the brand launches its own authorized generic? No one calls it a violation.
Industry insiders say the window for profitable first-generic entry has shrunk to 45-60 days in many categories. Companies are now building "dual-path" strategies: launching generics faster, diversifying portfolios, and even partnering with brand companies to get access to authorized generic distribution-just to survive.
What’s Next?
By 2027, authorized generics could make up 25-30% of all generic prescriptions, up from 18% in 2022. That means more drugs will be sold under generic labels-but still controlled by the same companies that made the brand.
The system is no longer about who can make the drug cheapest. It’s about who controls the timing. And until that changes, the promise of generic competition-lower prices, real choice, fair play-will keep getting delayed.
Can a brand company legally launch an authorized generic before the patent expires?
Yes. Authorized generics don’t need to wait for patent expiration because they’re sold under the brand’s existing New Drug Application (NDA). The FDA allows this as long as the product is identical and the brand authorizes the sale. This is different from traditional generics, which must wait until patent or exclusivity rights expire before filing an ANDA.
Why doesn’t the FDA stop authorized generics from entering during a first generic’s exclusivity period?
The FDA doesn’t regulate timing of market entry for authorized generics because they’re not considered a new drug application. Since they’re made under the brand’s NDA, the agency treats them as a marketing decision-not a competitive one. The regulatory framework simply doesn’t have rules to prevent this maneuver.
Do authorized generics cost less than traditional generics?
Not necessarily. Authorized generics often cost the same as traditional generics because they’re sold at similar wholesale prices. But because they enter the market early and split demand, they prevent the deep price drops that happen when multiple generics compete. So while the sticker price may look similar, the overall market savings are reduced.
How does the 180-day exclusivity period work?
The 180-day exclusivity period is granted to the first company to file a successful ANDA challenging a brand-name drug’s patent. During this time, no other generic can enter the market. But this protection doesn’t apply to authorized generics, which are not considered "generic competitors" under the law. So even if a first generic has exclusivity, a brand company can still launch its own version and legally share the market.
Are authorized generics safer or more reliable than traditional generics?
They’re identical in quality, safety, and effectiveness. Both must meet FDA standards. The only difference is who makes them. Authorized generics are made in the same facility as the brand drug, so some patients and pharmacists prefer them. But traditional generics are just as safe-they’re just made by a different company after going through the full ANDA process.


Jesse Hall
March 23, 2026 AT 21:12Man, this is wild. I didn’t realize the system was this rigged. I’ve been on Copaxone for years, and when the generic hit, I thought I was saving cash. Turns out my pharmacy was just selling Pfizer’s own version under a different label. 😅
Feels like they’re playing chess while we’re stuck playing checkers.
Donna Fogelsong
March 25, 2026 AT 01:32Big Pharma owns the FDA. It’s all connected. Same people. Same lobbyists. Same backroom deals. The 180-day exclusivity? A joke. They let you win the battle so they can steal the war. And don’t get me started on how the Inflation Reduction Act is just theater. They’re laughing all the way to the bank while we pay $150 for a 30-day supply of something that should cost $12.
Sean Bechtelheimer
March 25, 2026 AT 22:05They’re not even hiding it anymore. Authorized generics? More like authorized monopolies. 🤡
Remember when the FDA said they were 'identical'? Yeah, right. Identical until you check the manufacturing logs. Brand companies use the same factory, same equipment, same workers-then slap a 'generic' label on it and call it competition. It’s a scam dressed in white coats.
And the FTC? They’re asleep. Or worse-they’re on payroll.
Seth Eugenne
March 27, 2026 AT 16:44This is such a heartbreaking breakdown. I work in community pharmacy, and I see this every day. Patients come in thrilled they found a 'generic'-only to find out it’s still the same price as the brand because the market was split before real competition could kick in.
It’s not just about money. It’s about trust. When people think they’re getting a fair deal and then realize they were manipulated… that’s the real cost.
Small generic makers? They’re not just losing revenue-they’re losing hope. I’ve seen clinics shut down because they couldn’t afford to keep stocking generics that never dropped in price.
We need real reform. Not just talking points. Real teeth.
Caroline Dennis
March 29, 2026 AT 10:49Exclusivity without enforcement is a loophole with teeth. The Hatch-Waxman Act was designed to incentivize challenge, not enable co-optation. The authorized generic is a regulatory arbitrage-exploiting a structural blind spot.
It’s not innovation. It’s extraction.
Raphael Schwartz
March 30, 2026 AT 14:32fake generics are just a way for big pharma to keep prices high. america is getting screwed. why dont we just ban this crap? we dont need these corporate tricks. make em pay. #drugpricecrisis
winnipeg whitegloves
March 30, 2026 AT 18:48As a Canadian, I’m baffled how the U.S. lets this slide. Down here, if a brand wants to sell a generic version, they have to play by the same rules as everyone else-no sneaky backdoor launches. We don’t let them use their own patent monopoly as a weapon against competition.
It’s like letting the referee also be the team captain. The whole system’s got a glitch in its code. Someone needs to hit reset.
Marissa Staples
March 31, 2026 AT 05:29It’s funny how we call it 'competition' when it’s really just the same company wearing a different hat. The real tragedy? We’re all just tired. Too tired to fight it. Too tired to care. And that’s how they win.