90DayMeds.com: Your Ultimate Pharmaceuticals Resource - Page 2

Essential Tremor: Understanding the Disorder and Beta-Blocker Treatment Options
Explore the link between Essential Tremor and beta-blocker therapy, including propranolol dosing, side effects, and alternative treatments for managing movement disorder symptoms.
March 31 2026
How to Safely Restart Medication After a Break Without Overdose
Learn why restarting medication after a break carries severe overdose risks due to lost tolerance. Discover safe dosing strategies, high-risk drug classes, and essential prevention tools.
March 26 2026
Anxiety and Nervousness Caused by Medications: Triggers and Solutions
Certain medications like steroids, ADHD drugs, and thyroid meds can trigger anxiety symptoms. Learn which drugs cause it, how to tell if it's the pill or a mental health issue, and what steps to take to feel better.
March 24 2026
First Generic vs Authorized Generic: Timing of Market Entry in U.S. Drug Market
First generics and authorized generics enter the market differently, with timing that affects drug prices. While first generics wait years to challenge patents, brand companies can launch authorized versions instantly-undercutting competition and slowing price drops.
March 23 2026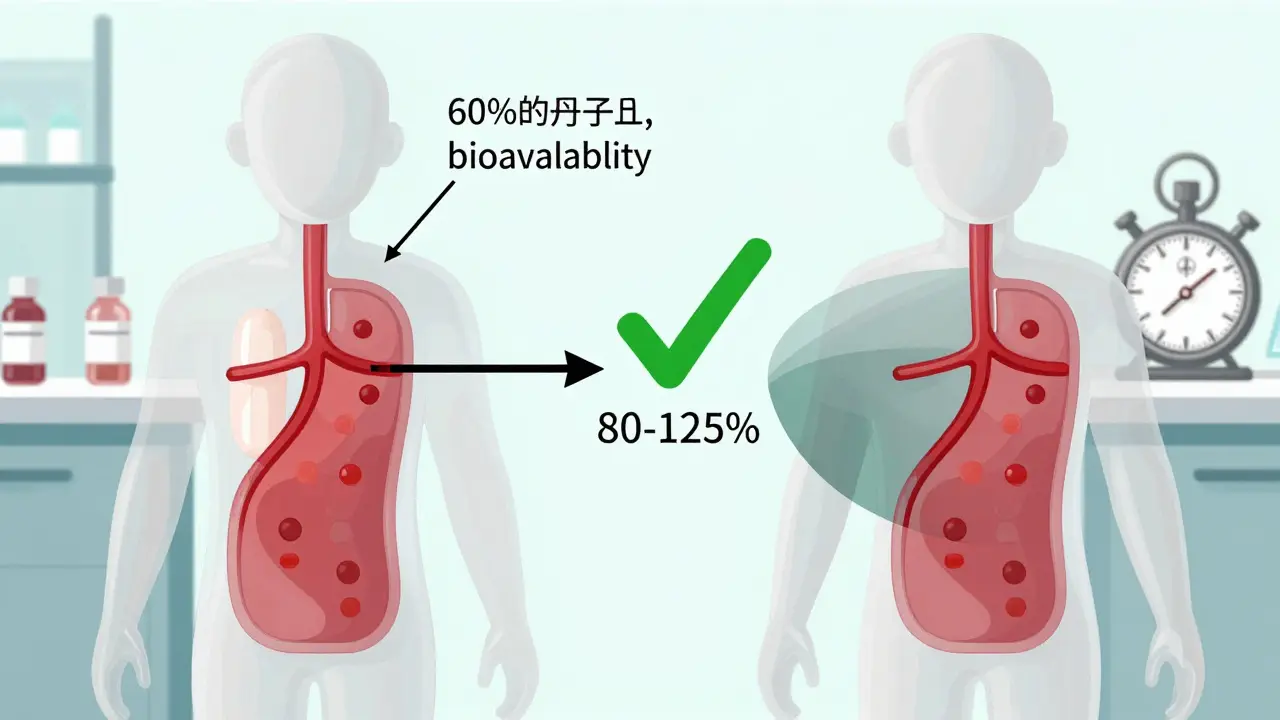
How to Compare Bioavailability and Bioequivalence in Generic vs Brand Drugs
Bioavailability measures how much of a drug enters your bloodstream, while bioequivalence proves a generic matches the brand. Learn how regulators ensure generics work just as well - and when exceptions apply.
March 17 2026
Combination NTI Drugs and Generic Availability: Coverage and Gaps
Combination NTI drugs offer critical treatment for complex conditions, but generic versions are nearly nonexistent due to strict bioequivalence challenges. Patients face higher risks of failure and toxicity when substitutions occur.
March 12 2026
Continuing Education for Pharmacists: Staying Current on Generics
Pharmacists must stay current on generics to ensure safe substitutions. With 90% of prescriptions filled as generics, understanding therapeutic equivalence, state laws, and FDA updates is critical for patient safety and legal compliance.
March 10 2026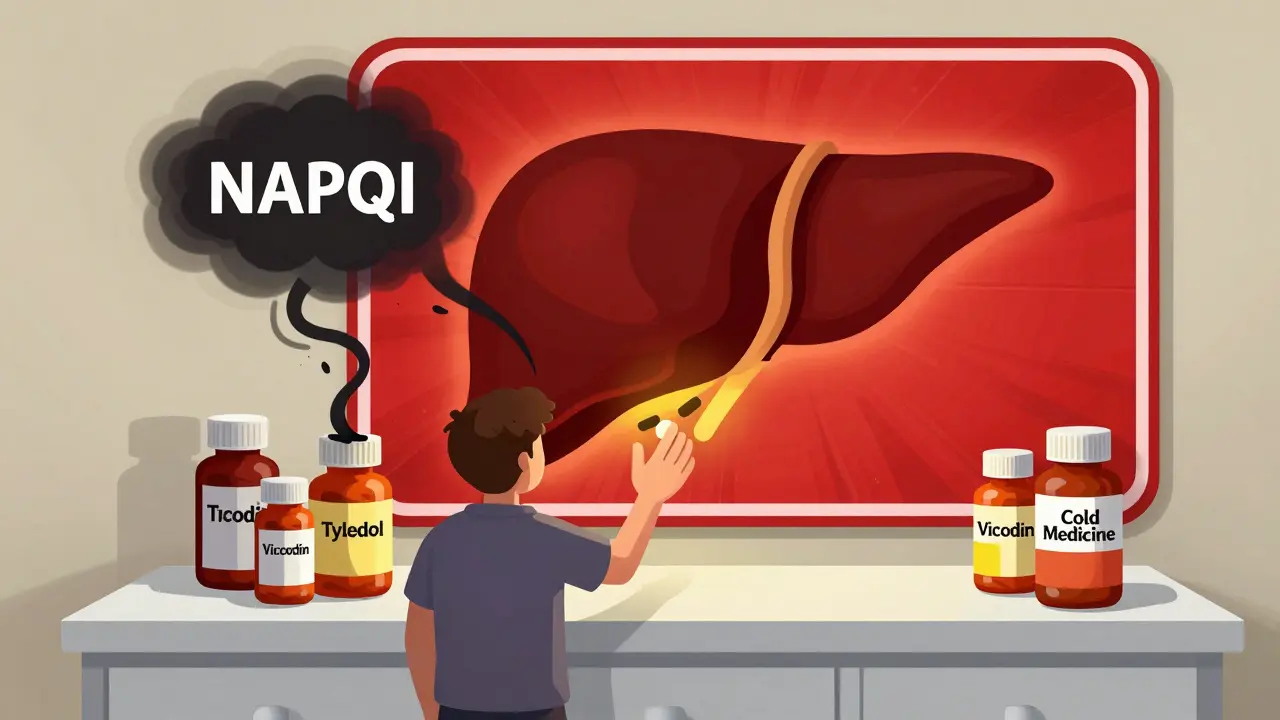
Acetaminophen Overdose: Signs of Liver Damage and What to Do
Acetaminophen overdose is the top cause of acute liver failure in the U.S. Learn the hidden signs, how NAC saves lives, and why even one extra pill can be deadly. Timing is everything.
March 9 2026
Generic Drug Recalls: Why They Happen and What to Do
Generic drug recalls are more common than you think, often due to manufacturing flaws. Learn why they happen, what the FDA classes mean, and exactly what to do if your medication is affected.
March 3 2026
Hyperglycemia: High Blood Sugar Symptoms and Emergency Management
Hyperglycemia is a dangerous spike in blood sugar that can lead to life-threatening emergencies like DKA and HHS. Learn the early signs, how to respond, and when to seek emergency care.
March 2 2026